Geologists, biologists unearth the atomic fingerprints of cancer
Scientists at the University of Colorado Boulder and Princeton University have, for the first time, employed a tool often used in geology to detect the atomic fingerprints of cancer.
Scientists at the University of Colorado Boulder and Princeton University have, for the first time, employed a tool often used in geology to detect the atomic fingerprints of cancer.
Biochemistry
9 hours ago
0
36

Planetary scientists at the University of Colorado Boulder have discovered how Venus, Earth's scalding and uninhabitable neighbor, became so dry.
Planetary Sciences
13 hours ago
0
39

Move over, graphene. There's a new, improved two-dimensional material in the lab. Borophene, the atomically thin version of boron first synthesized in 2015, is more conductive, thinner, lighter, stronger and more flexible ...
Nanomaterials
14 hours ago
0
34
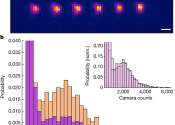
A team of physicists at Harvard University has succeeded in trapping individual polyatomic molecules in optical tweezer arrays for the first time. In their paper published in the journal Nature, the group describes how they ...

Scientists at the Department of Energy's Oak Ridge National Laboratory have developed lubricant additives that protect both water turbine equipment and the surrounding environment.
Analytical Chemistry
May 2, 2024
1
95

Proximity is key for many quantum phenomena, as interactions between atoms are stronger when the particles are close. In many quantum simulators, scientists arrange atoms as close together as possible to explore exotic states ...
Condensed Matter
May 2, 2024
2
168

For the first time, chemists in the University of Minnesota Twin Cities College of Science and Engineering have created a highly reactive chemical compound that has eluded scientists for more than 120 years. The discovery ...
Analytical Chemistry
May 1, 2024
0
63

The European Galileo navigation system has two more satellites in orbit following their launch in the early morning of Sunday, 28 April, at 01:34 BST/02:34 CEST. With 30 satellites now in orbit, Galileo is expanding its constellation, ...
Space Exploration
Apr 30, 2024
0
1

Creating heat from fusion reactions requires carefully manipulating the properties of plasma, the electrically charged fourth state of matter that makes up 99% of the visible universe.
Plasma Physics
Apr 30, 2024
0
41

Oxidative redispersion at elevated temperatures has long been utilized in heterogeneous catalysis for the regeneration of sintered metal catalysts and the synthesis of metal single atom and cluster catalysts. These redispersion ...
Nanomaterials
Apr 30, 2024
0
35
The atom is a basic unit of matter consisting of a dense, central nucleus surrounded by a cloud of negatively charged electrons. The atomic nucleus contains a mix of positively charged protons and electrically neutral neutrons (except in the case of hydrogen-1, which is the only stable nuclide with no neutron). The electrons of an atom are bound to the nucleus by the electromagnetic force. Likewise, a group of atoms can remain bound to each other, forming a molecule. An atom containing an equal number of protons and electrons is electrically neutral, otherwise it has a positive or negative charge and is an ion. An atom is classified according to the number of protons and neutrons in its nucleus: the number of protons determines the chemical element, and the number of neutrons determine the isotope of the element.
The name atom comes from the Greek ἄτομος/átomos, α-τεμνω, which means uncuttable, something that cannot be divided further. The concept of an atom as an indivisible component of matter was first proposed by early Indian and Greek philosophers. In the 17th and 18th centuries, chemists provided a physical basis for this idea by showing that certain substances could not be further broken down by chemical methods. During the late 19th and early 20th centuries, physicists discovered subatomic components and structure inside the atom, thereby demonstrating that the 'atom' was divisible. The principles of quantum mechanics were used to successfully model the atom.
Relative to everyday experience, atoms are minuscule objects with proportionately tiny masses. Atoms can only be observed individually using special instruments such as the scanning tunneling microscope. Over 99.9% of an atom's mass is concentrated in the nucleus, with protons and neutrons having roughly equal mass. Each element has at least one isotope with unstable nuclei that can undergo radioactive decay. This can result in a transmutation that changes the number of protons or neutrons in a nucleus. Electrons that are bound to atoms possess a set of stable energy levels, or orbitals, and can undergo transitions between them by absorbing or emitting photons that match the energy differences between the levels. The electrons determine the chemical properties of an element, and strongly influence an atom's magnetic properties.
This text uses material from Wikipedia, licensed under CC BY-SA