Single molecules show promise to optically detect single electrons
Optical detection of a single electron using a single molecule has never been done. Leiden physicist Michel Orrit and his team have now identified a molecule that is sensitive enough to detect an electron at a distance of hundreds of nanometers. The results are published as a cover article in ChemPhysChem.
Physicists have been able to manipulate single electrons for some time. But they can only see them as part of an electric current consisting of thousands of electrons. A goal in physics is a method of indirectly detecting individual electrons using a single molecule. In the future, a quantum computer could use this method to locate qubits with light without disturbing their spin quantum state—an essential requirement for quantum computers. Leiden physicist Michel Orrit and his group have now taken a first step toward developing this technique by identifying a molecular system that is sensitive enough to detect an electron from as far as hundreds of nanometers away.
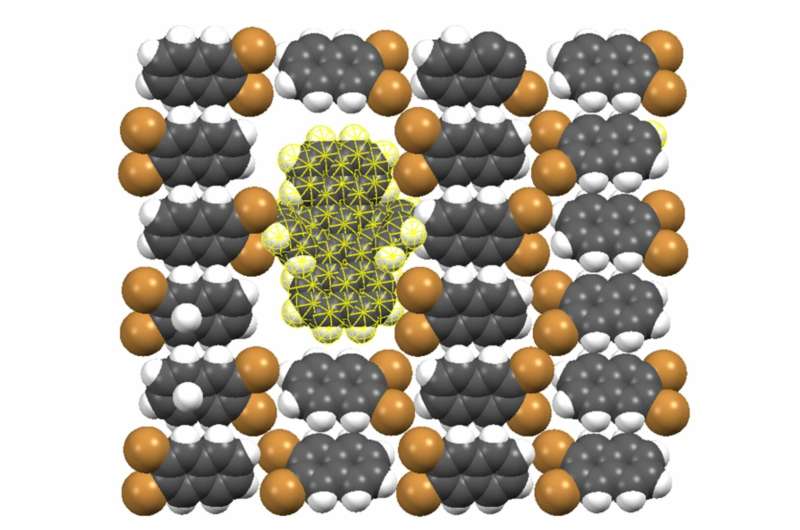
The researchers, including lead authors Zoran Ristanović and Amin Moradi, found that the fluorescent molecule dibenzoterrylene (DBT) possesses two vital properties for single charge detection—provided that it is included in a molecular crystal of 2,3-dibromonaphthalene. First, DBT molecules fluoresce, emitting a narrow spectrum of visible light that is stable over longer periods of time (fig. 1). Second, those narrow spectral lines shift significantly in the presence of an electric field (fig. 2). This will become the telltale sign of a nearby charge, because charges generate such an electric field.
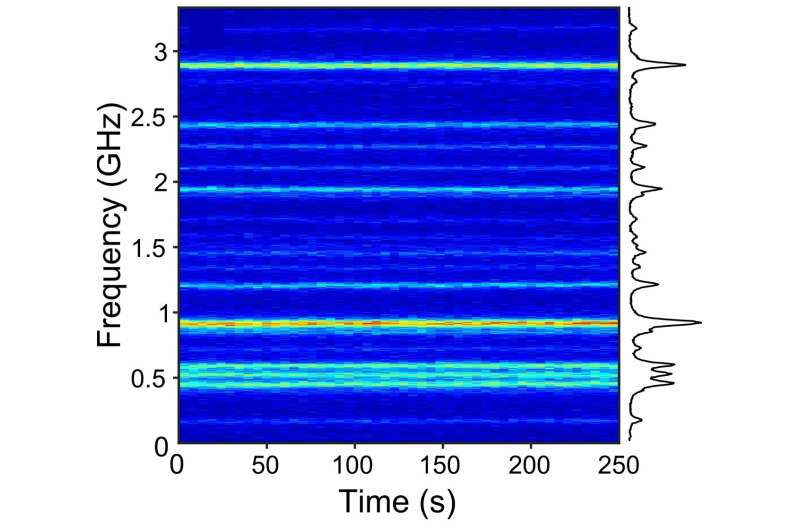
Orrit and his colleagues show that they can easily detect electric fields in the order of 1 kV/cm (fig. 2) with a DBT-molecule. This is more than enough sensitivity for detecting a single electron at 100 nm away, whose electric field is about 1.5 kV/cm. Using multiple molecules responding similarly to an electric field, the physicists could even use triangulation to find the electron's location, similar to GPS. The next step is detecting an actual electron. The research team is currently building a single-electron device for that experiment.
More information: Amin Moradi et al. Cover Feature: Matrix-induced Linear Stark Effect of Single Dibenzoterrylene Molecules in 2,3-Dibromonaphthalene Crystal (ChemPhysChem 1/2019), ChemPhysChem (2018). DOI: 10.1002/cphc.201801157
Journal information: ChemPhysChem
Provided by Leiden Institute of Physics