January 3, 2019 feature
Glassy carbon microneedles: A new transdermal drug delivery device
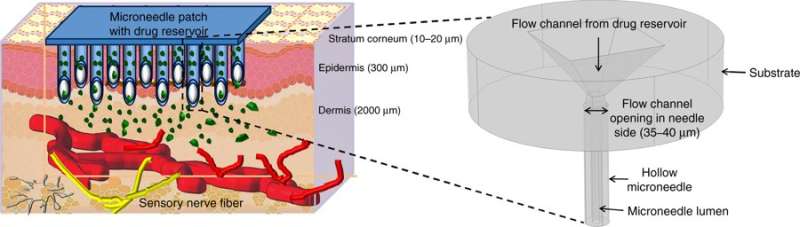
Microneedles are designed to infuse medications transdermally (through the skin) and relatively painlessly during clinical applications. In a recent study, Richa Mishra and her colleagues developed a new technique to convert polymer films into a fracture-resistant glassy form of carbon to develop microneedles. The optimized transdermal drug delivery device was fabricated using photolithography to form a biocompatible and robust microneedle for pre-clinical applications.
The scientists first generated an array of vertical, hollow tubes made from the epoxy-based polymer SU-8 on a silicon substrate. The microstructures were sharpened into needle-like shapes by heat treatment in an oxygen-free oven and the final product was composed entirely of randomly ordered carbon atoms. Subsequent materials characterization included nanoindentation to measure the surface stiffness. The glassy carbon structures were robust enough to penetrate human skin without breaking. Results of the study were published on Microsystems & Nanoengineering.
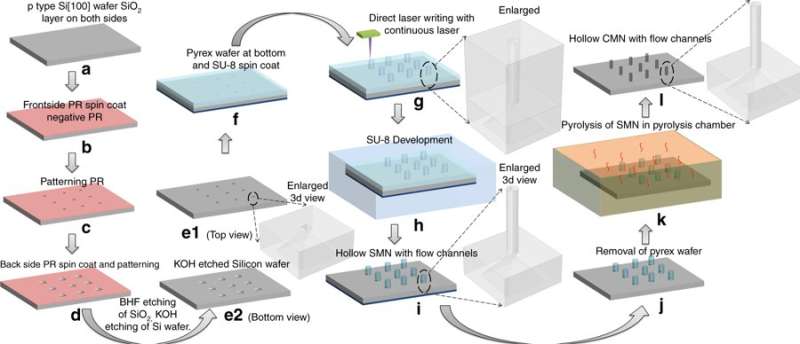
The authors detailed the conventional carbon-microelectromechanical system (C-MEMS) process used to fabricate the microneedles. In the process, Mishra et al. used a scalable and irreversible step of pyrolysis, where the precursor of pre-patterned SU-8 microneedles were converted to glassy carbon structures. The experiments were conducted in an inert atmosphere at high temperatures (~900 0C) to retain their original shape after shrinking. On conversion to glassy carbon, the microneedles inherited the associated properties of hardness, biocompatibility, thermal and chemical resistance.
The researchers compared the hardness and Young's modulus of the carbon and SU-8 microneedles to understand increased strength induced by the C-MEMS process. Structural shrinking of carbon microneedles on pyrolysis was estimated thereafter. Mishra et al. conducted in-depth materials characterization with energy-dispersive X-ray spectroscopy (EDX) to understand surface chemistry and Raman spectroscopy to estimate the atomic percentage of carbon in the microneedle's crystalline structure.
The results confirmed the glassy composition of the microneedles. Additional compression and bending tests determined the maximum forces the microneedles could withstand. The devices could withstand forces that were approximately two orders of magnitude higher than resistive forces of the skin. The devices were subsequently tested on the skin surface of a mouse model in the study.
The use of carbon in medicine is not new; examples include applications in orthopedic joints, as carbon fibres/composites in orthopedic surgeries and as materials in surgical instruments. The properties of carbon fibres to form 3-D scaffold-like structures has shown promising capacity in bone tissue regeneration. The tailored properties of carbon have made them suitable to fabricate transdermal drug delivery microneedles (MN) for advanced drug and vaccine delivery.
For competence, MNs should have an optimal length for effective drug delivery, but be short enough to avoid causing pain. Materials scientists are exploring strategies to select clinically viable materials and methods that can be commercially translated in a short period of time.
The key requirements for MNs include biocompatibility, strength and controlled drug delivery. Solid MNs can be developed with polymers, silicon, steel, titanium and nickel via subtractive or additive processes. One disadvantage of solid MNs is their limited capacity for optimal drug delivery. On the other hand, hollow MNs can be attached to a drug reservoir with external pressure for optimal drug delivery.
Mishra et al. chose the precursor SU-8 for its high cross-linking strength, biocompatibility, low-cost, light-induced polymerization and compatibility with the microelectronic industry process. To fabricate hollow, glassy carbon microneedles (CMNs), the C-MEMS process was first adapted to pattern SU-8 microneedles (SMNs) using direct laser writing, followed by their conversion to glassy CMNs via pyrolysis. Microfluidic ports were etched in a silicon wafer, according to a previous protocol developed by the same research team. The resulting products (CMNs) were characterized for their mechanical properties, including hardness and Young's modulus in comparison with their precursor SMNs. The results indicated that CMNs were superior.
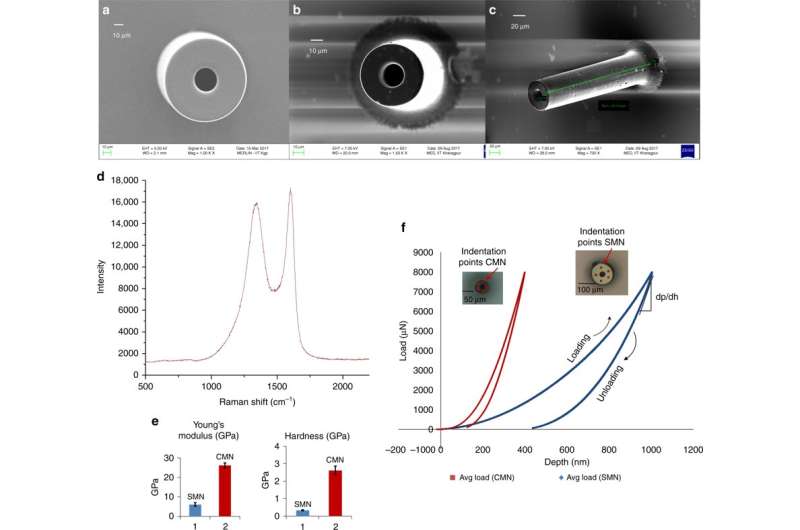
Mishra et al. observed and quantified the dimensions of SMN precursors and corresponding pyrolyzed CMN structures using scanning electron microscopy (SEM) images. After pyrolysis, the diameter and wall thickness of the microneedle decreased. The EDX results showed increased atomic percentage of carbon elements for carbon MNs. The absence of nitrogen in the carbon microneedle's composition indicated its removal during heating. The percent of oxygen content was due to the CMN structure and the Si/SiO2 substrate surface. Raman spectroscopy showed the glassy crystalline nature of the final microstructure by means of verifying the carbon product. The results agreed with previous studies.
The scientists obtained load displacement data via nanoindentation for the separate SU-8 and carbon structures. They calculated the hardness and modulus of elasticity using the Oliver Pharr model. The results showed a hardness of 0.33 GPa and Young's modulus of 5.52 GPa for SMN. On conversion to CMN via pyrolysis, the indentation results showed higher elasticity of the material, the hardness increased eight-fold to 2.62 GPa and the Young's modulus increased 4.8-fold to 26.97 GPa.
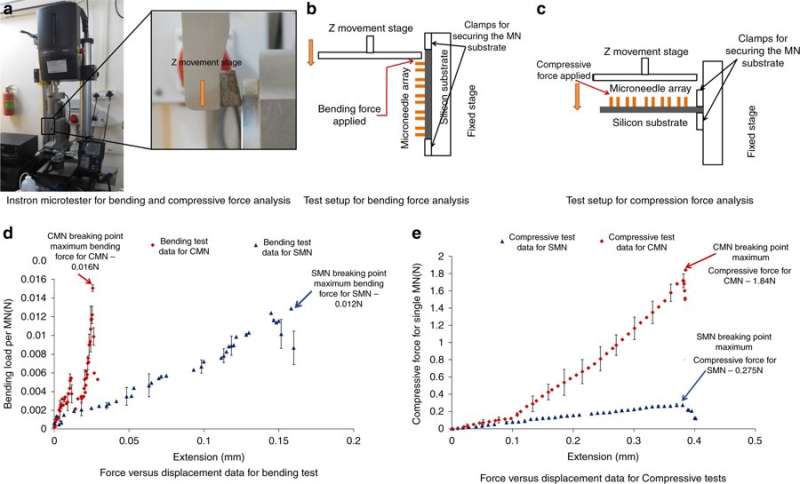
To understand the capacity of MNs to withstand forces without breakage during insertion into the skin, Mishra et al. conducted bending and compression force measurements of the structure. Typically, an MN experiences resistance during skin penetration, the applied force should therefore be greater than the opposing force. To test bending/compression, the MNs were loaded to an Instron micro-tester and a metal plate was driven toward the MNs until they broke; if the applied load was below the maximum compressive or bending force, the needles will not break. A sharp drop in the compressive force or bending force indicated a fracture point. The results showed that CMNs were much stronger than the precursors, allowing them to overcome resistance during skin penetration.
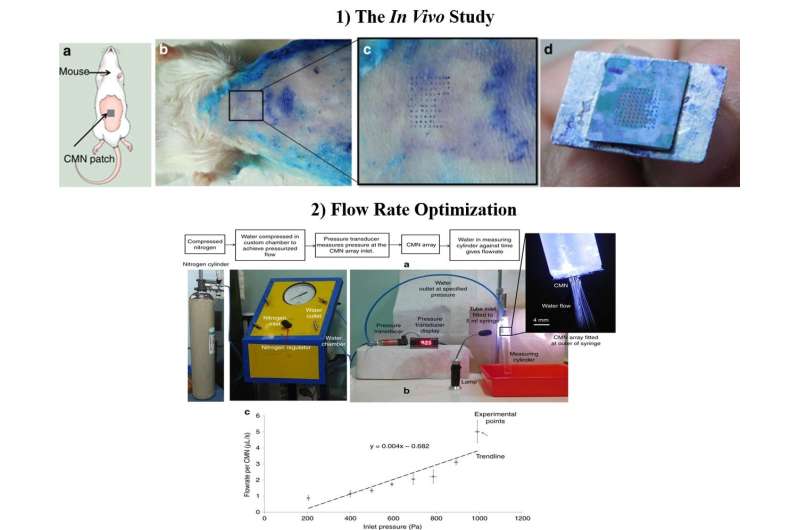
After confirming the mechanical character and superiority of hollow CMNs in the lab, Richa Mishra and co-workers conducted in vivo experiments to test the CMNs on mouse skin. A microneedle array freshly dipped in methylene blue was pressed on to the skin of a Swiss Albino mouse model and slowly withdrawn multiple times. The needle marks of methylene blue were visible, the MNs remained intact after multiple insertions and a 500 µm array spacing was optimal for further studies in the lab.
The scientists optimized flow dimensions during the process of manufacture and included microfluidic conduits with reduced size for enhanced MN functionality. To test the flow rates, they attached a 5-mL syringe with a customized chamber containing deionized water. When the scientists measured the average flow rate versus pressure for individual MNs, the results showed that drug delivery could be ultimately controlled by modifying the inlet pressure.
The definitive materials characterization technique in the study was nanoindentation, since the parameter quantified surface hardness and Young's modulus to verify the superiority of CMNs. Future work will include the development of controlled drug delivery, with a micropump and a drug reservoir integrated to the MN design. In this way, the scientists aim to develop advanced drug and vaccine delivery systems in healthcare that are cost-efficient, accurate and painless to the patient.
More information: Richa Mishra et al. Glassy carbon microneedles—new transdermal drug delivery device derived from a scalable C-MEMS process, Microsystems & Nanoengineering (2018). DOI: 10.1038/s41378-018-0039-9
S.A. Catledge et al. Nanostructured diamond coatings for orthopaedic applications, Diamond-Based Materials for Biomedical Applications (2013). DOI: 10.1533/9780857093516.2.105
W.C. Oliver et al. An improved technique for determining hardness and elastic modulus using load and displacement sensing indentation experiments, Journal of Materials Research (2007). DOI: 10.1557/JMR.1992.1564
Mark R. Prausnitz et al. Current status and future potential of transdermal drug delivery, Nature Reviews Drug Discovery (2004). DOI: 10.1038/nrd1304
Journal information: Nature Reviews Drug Discovery
© 2019 Science X Network




















