Turbulence allows clinical-scale platelet production for transfusions
Turbulence is a critical physical factor that promotes the large-scale production of functional platelets from human induced pluripotent stem cells (hiPSCs), researchers in Japan report July 12 in the journal Cell. Exposure to turbulent energy in a bioreactor stimulated hiPSC-derived bone marrow cells called megakaryocytes to produce 100 billion platelets—blood cell fragments that help wounds heal and prevent bleeding by forming blood clots. Moreover, transfusion of these platelets in two animal models promoted blood clotting and prevented bleeding just as well as human donor platelets.
"The discovery of turbulent energy provides a new physical mechanism and ex vivo production strategy for the generation of platelets that should impact clinical-scale cell therapies for regenerative medicine," says senior study author Koji Eto, part of the Center for iPS Cell Research and Application at Kyoto University.
Blood transfusion is one the most common forms of cell therapy, with nearly five million Americans undergoing this procedure each year. In the near future, donor blood supplies are not expected to meet patient demand in several countries. One factor that contributes to this problem is the short shelf-life of some blood components.
In particular, human donor platelets have a shelf life of only 5 days in the United States because they gradually lose their aggregation capacity and are susceptible to bacterial contamination. Platelet transfusions are sometimes needed to treat a condition called thrombocytopenia, in which platelet deficiency increases the risk of life-threatening blood loss. The expected shortage of platelets has stimulated researchers to look for alternative sources that don't rely on blood donations.
hiPSCs offer a renewable approach for producing sufficient numbers of platelets for transfusion. This technique involves epigenetically reprogramming blood or skin cells taken from human donors to an embryonic-stem-cell-like state and then converting these immature cells into specialized cell types found in different parts of the body. However, previous attempts to generate platelets from hiPSC-derived megakaryocytes have failed to achieve a scale suitable for clinical manufacturing.
While searching for a solution to this problem, Eto and his collaborators noticed that hiPSC-derived megakaryocytes produced more platelets when being rotated in a flask than under static conditions in a petri dish. This observation suggested that that physical stress from horizontal shaking under liquid conditions enhances platelet generation. Following up on this discovery, the researchers tested a rocking-bag-based bioreactor followed by a new microfluidic system with a flow chamber and multiple pillars, but these devices generated fewer than 20 platelets per hiPSC-derived megakaryocyte.
To examine the ideal physical conditions for generating platelets, Eto and his team next conducted live-imaging studies of mouse bone marrow—the tissue that produces blood components. These experiments revealed that megakaryocytes release platelets only when they are exposed to turbulent blood flow. In support of this idea, simulations confirmed that the bioreactor and microfluidic system they previously tested lacked sufficient turbulent energy.
"The discovery of the crucial role of turbulence in platelet production significantly extends past research showing that shear stress from blood flow is also a key physical factor in this process," Eto says. "Our findings also show that iPS cells are not the end-all be-all for producing platelets. Understanding fluid dynamics in addition to iPS cell technology was necessary for our discovery."
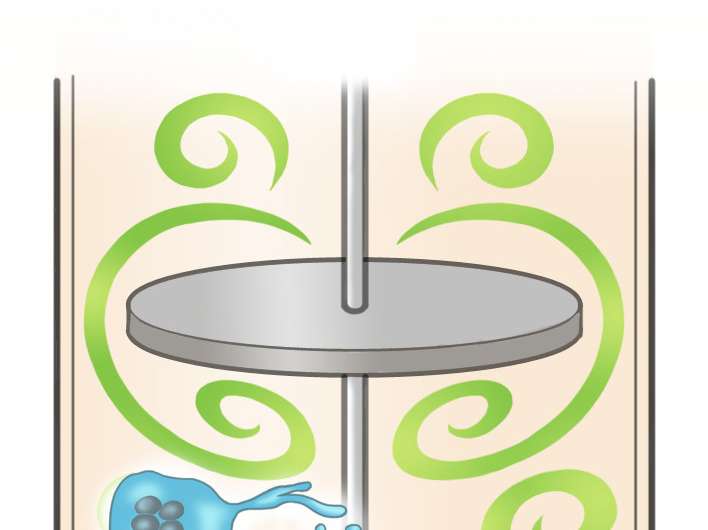
After thoroughly testing various devices, the researchers discovered that large-scale production of high-quality platelets was possible using a bioreactor called VerMES. This system consists of two oval-shaped, horizontally oriented mixing blades that generate relatively high levels of turbulence by moving up and down in a cylinder. With the optimal level of turbulent energy and shear stress created by the blade motion, the hiPSC-derived megakaryocytes generated 100 billion platelets—enough to satisfy clinical requirements.
Transfusion experiments in two animal models with thrombocytopenia showed that these platelets perform similarly to human donor platelets. Specifically, both types of platelets promoted blood clotting and reduced bleeding times to a comparable extent after ear vein incisions in rabbits and tail artery punctures in mice.
Currently, Eto and his team are improving their approach by designing automated protocols, lowering manufacturing costs, and optimizing platelet yields. They are also developing universal platelets lacking cell-surface proteins called human leukocyte antigens in order to reduce the risk of immune-mediated transfusion reactions.
"We expect clinical trials to begin within a year or two," Eto says. "We believe these findings will be a last scientific step to receiving permission for clinical trials using our platelets."
More information: Cell, Ito and Nakamura et al.: "Turbulence activates platelet biogenesis to enable clinical scale ex vivo production" www.cell.com/cell/fulltext/S0092-8674(18)30736-0 , DOI: 10.1016/j.cell.2018.06.011
Journal information: Cell
Provided by Cell Press