Beef peptides block bitter tastes
From burgers to steaks, beef has a long history of being a delicious part of dinner. But what if that pleasant experience of eating beef could extend beyond the dinner plate? Now, one group reports in ACS' Journal of Agricultural and Food Chemistry that beef protein, when broken down into peptides, can block bitter taste receptors on the tongue. Such peptides could someday be used to make other foods and even medicines taste better.
Most people try to avoid bitter flavors because they find them to be unpleasant. But some healthful foods are bitter, as are some medications. So, the food and pharmaceutical industries have been looking at ways to reduce or eliminate bitter sensations, which are detected in humans by 25 receptors known as T2Rs. Only a few inhibitors of T2R activity have been identified so far. In recent years, bioactive peptides created from breaking down food proteins, through a process known as enzymatic hydrolysis, have gained attention for reducing bitterness and inflammation. Because beef proteins have been shown to generate desirable flavor-promoting peptides, Prashen Chelikani, Rotimi E. Aluko and colleagues wanted to see if these peptides could block bitter tastes.
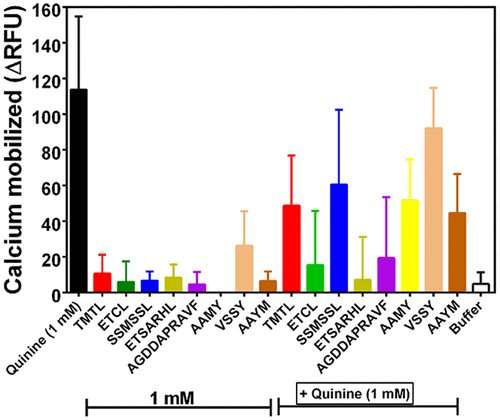
The researchers hydrolyzed beef protein with six different enzymes: alcalase, chymotrypsin, trypsin, pepsin, flavourzyme and thermoase. Peptides produced from trypsin and pepsin digestion were the most effective in reducing the intensity of the bitterness of quinine in a test with an electronic tongue. These peptides were also the longest, which suggests that peptide size might play an important role. The group notes this could impact not only the food industry but the pharmaceutical industry as well.
More information: Chunlei Zhang et al. Beef Protein-Derived Peptides as Bitter Taste Receptor T2R4 Blockers, Journal of Agricultural and Food Chemistry (2018). DOI: 10.1021/acs.jafc.8b00830
Abstract
The aim of this work was to determine the T2R4 bitter taste receptor-blocking ability of enzymatic beef protein hydrolysates and identified peptide sequences. Beef protein was hydrolyzed with each of six commercial enzymes (alcalase, chymotrypsin, trypsin, pepsin, flavourzyme, and thermoase). Electronic tongue measurements showed that the hydrolysates had significantly (p < 0.05) lower bitter scores than quinine. Addition of the hydrolysates to quinine led to reduced bitterness intensity of quinine with trypsin and pepsin hydrolysates being the most effective. Addition of the hydrolysates to HEK293T cells that heterologously express one of the bitter taste receptors (T2R4) showed alcalase, thermoase, pepsin, and trypsin hydrolysates as the most effective in reducing calcium mobilization. Eight peptides that were identified from the alcalase and chymotrypsin hydrolysates also suppressed quinine-dependent calcium release from T2R4 with AGDDAPRAVF and ETSARHL being the most effective. We conclude that short peptide lengths or the presence of multiple serine residues may not be desirable structural requirements for blocking quinine-dependent T2R4 activation.
Journal information: Journal of Agricultural and Food Chemistry
Provided by American Chemical Society


















