Scientists discover a link between superconductivity and the periodic table
Scientists from Moscow Institute of Physics and Technology and Skoltech have demonstrated the high-temperature superconductivity of actinium hydrides and discovered a general principle for calculating the superconductivity of hydrides based on the periodic table alone. The results of their study were published in the Journal of Physical Chemistry Letters.
High-temperature superconductivity is a phenomenon of zero electrical resistance in certain materials at temperatures above -196 degrees C (the temperature of liquid nitrogen). Room-temperature superconductors open up prospects for the power industry, transport, and other technology-driven sectors. Currently, the record holder in high-temperature superconductivity is hydrogen sulfide (H3S), which functions as a superconductor at 1.5 million atmospheres and a temperature of -70 degrees C. Such pressure levels can only be attained in a lab environment, and the temperature is way below room temperature, so the search continues for new superconductors. Perhaps an even higher-temperature superconductivity can be attained in metal-hydrogen compounds. Yet the link between chemical composition and superconductivity was unclear, leaving scientists to puzzle out by trial and error.
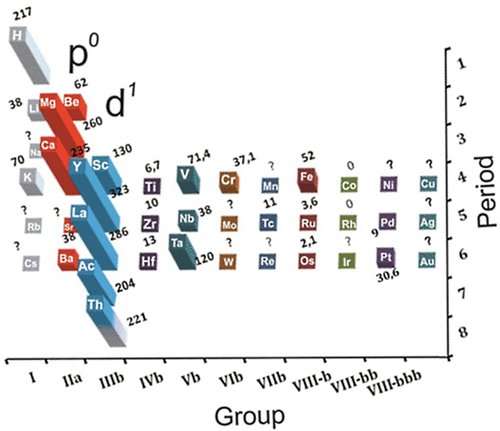
A group of chemists led by Artem R. Oganov, professor at the Moscow Institute of Physics and Technology and Skoltech, discovered that certain elements capable of forming superconducting compounds are arranged in a specific pattern in the periodic table. It was established that high-temperature superconductivity develops in substances containing metal atoms that come close to populating a new electronic subshell. Metal atoms inside the crystal are assumed to become highly sensitive to the positions of the neighboring atoms, which would result in strong electron-phonon interaction, the underlying effect of conventional superconductivity. Based on this assumption, the scientists supposed that high-temperature superconductivity could occur in actinium hydrides. Their supposition was verified and confirmed: superconductivity was predicted for AcH16 at temperatures of -69 to -22 degrees C at 1.5 million atmospheres.
"The very idea of a connection between superconductivity and the periodic table was first put forward by Dmitry Semenok, a student at my lab. The principle he discovered is very simple and it is really amazing that no one had hit upon it before," says Artem Oganov.
More information: Dmitrii V. Semenok et al, Actinium Hydrides AcH10, AcH12, and AcH16 as High-Temperature Conventional Superconductors, Journal of Physical Chemistry Letters (2018). DOI: 10.1021/acs.jpclett.8b00615
Journal information: Journal of Physical Chemistry Letters
Provided by Moscow Institute of Physics and Technology