Scientist describes fundamental process when ice is compressed
Almost three-quarters of the earth's surface is covered by water. Almost two-thirds of the human body is made up of it. We drink it. We use it in our homes and in industry. As a solid, it's ice. As a gas, it's steam.
"Nobody understands water, the structure of water. Water has a lot of anomalies," says John Tse, University of Saskatchewan physics professor and Canada Research Chair in Materials Science.
Tse has committed decades to learning more about what so many of us take for granted. In the process, he has refuted a long-standing fundamental hypothesis about water. The results were published this fall in Physical Review Letters.
Curious about water and how it behaves when compressed to a solid, Tse was interested in an experiment conducted in 1985 by some of his colleagues when he worked at the National Research Council. They discovered an unusual phenomenon. When they compressed ice at low temperatures, instead of transforming into a high-pressure crystalline form where the atoms are organized into a lattice pattern, the ice converted to an amorphous solid and the atoms were disorganized. They hypothesized that this was due to "melting" of ice at high pressure. If that were the case, the disordered state would be water-like.
These conclusions didn't sit well with Tse because the consequence would be that liquid water is a mixture of two liquids with different densities. At the time, however, neither the technology nor the expertise was available to test the hypothesis.
"The question nobody asked was whether the hypothesis was correct," he says.
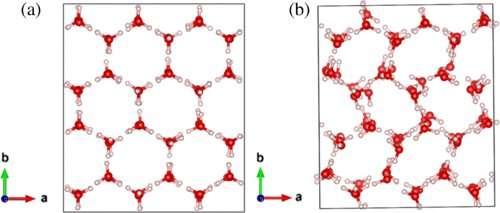
Thirty years later, Tse and a research team used two beamlines at the Advanced Photon Source, one of which is managed by the Canadian Light Source at the U of S. In his paper, "Kinetically Controlled Two-Step Amorphization and Amorphous-Amorphous Transition in Ice," Tse reports that the "water-like" state was actually an intermediate crystalline phase and not liquid.
"This changes anything to do with water," says Tse.
"We described how the ice can change from one form to another. That's the significance of the result, the reason that this is so important. We mapped out the phase diagram in the amorphous phase. It is a guide to tell you what are the properties of the matter at a certain pressure-temperature condition."
Science is driven either by curiosity or necessity or both, says Tse.
"Fundamental science is important work. If we don't work on fundamental science, we won't know where we come from. We will be missing something. Curiosity drives this research. We had a fundamental hypothesis and we did the experiment and tried to clarify. We now know exactly what is going on."
Tse continues his work on water and how it changes as a function of pressure and temperature. Water is even more complicated because the atoms are moving. In ice, the atoms are not moving but they are disordered. In both cases, he says, there is still more to learn.
"You always look at old problems that have not been resolved, but with the development a new instrument and a new theory, you might be able to solve the old problem and then build on it."
More information: Chuanlong Lin et al. Kinetically Controlled Two-Step Amorphization and Amorphous-Amorphous Transition in Ice, Physical Review Letters (2017). DOI: 10.1103/PhysRevLett.119.135701
Journal information: Physical Review Letters
Provided by Canadian Light Source