How ribosomes shape the proteome
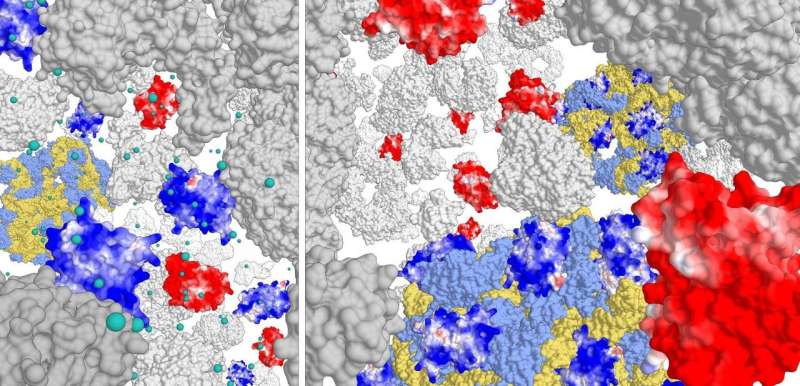
Cells are crowded with macromolecules, which limits the diffusion of proteins, especially in prokaryotic cells without active transport in the cytoplasm. While investigating the relationship between crowding, ionic strength and protein diffusion, University of Groningen biochemists made a fascinating discovery: positively charged proteins stick to the surface of ribosome complexes. This explains why most water-soluble proteins carry an overall negative charge. The discovery will appear soon in the journal eLife.
The speed of movement of proteins inside cells is important: many processes in biological cells depend on interactions between macromolecules (proteins and nucleic acids) and thus on their ability to find each other. 'But the cell cytoplasm is a bustling place and this will affect protein and RNA diffusion', remarks University of Groningen Professor of Biochemistry Bert Poolman.
Charges
His group studied the effects of crowding on diffusion, and found a correlation between protein size and diffusion speed. 'But for some proteins we did not find this correlation, so we set out to investigate why.' The team used three different prokaryotes with increasing ionic strength: the Gram-negative bacterium Escherichia coli, the Gram-positive Lactococcus lactis and the extremophile Haloferax volcanii, which lives at very high salt concentrations.
For this study, the researchers constructed different variants of Green Fluorescent Protein (GFP), with surface charges ranging from -30 to +25. They then studied the movement of these GFP variants in the three cell types. 'We saw that positively charged proteins would diffuse very slowly. They got stuck in the cell', explains Poolman. Further analysis showed that the positive proteins didn't bind to the DNA or the cell membrane but to the ribosome complex.
Interesting
A bioinformatics analysis of the proteomes of microorganisms and eukaryotic cells showed that in most cases roughly 70 percent of the proteins are negatively charged. 'Interestingly, the remaining 30 percent are either membrane proteins or proteins involved in the functioning or folding of the ribosome or mRNA.'
The membrane proteins are shielded by chaperones during biogenesis, so they won't stick to the ribosomes. There are therefore no 'free' cytoplasmic proteins with a high enough positive charge to make them settle onto ribosomes. The negative charge of the ribosome complex and the ambient ionic strength of the cytoplasm appear to have shaped the evolution of charges in the cellular proteome.
Unexpected
The new and unexpected insight that protein mobility is a function of protein charge may explain why it is hard to express some proteins in bacterial systems with low ionic strength. 'We observed that a higher ionic strength reduces the stickiness of positively charged proteins. That could be a valuable insight for the construction of protein expression platforms.'
A final observation in the eLife paper is that the genomes of several endosymbionts show an abundance of positively charged proteins. 'This finding really baffles us', admits Poolman. 'You would expect all these proteins to be attracted to the endosymbionts ribosomes. So far, we have no explanation of how these organisms are able to deal with slow diffusion and ribosomes being engulfed with positive proteins.'
More information: Paul E Schavemaker et al, Ribosome surface properties may impose limits on the nature of the cytoplasmic proteome, eLife (2017). DOI: 10.7554/eLife.30084
Journal information: eLife
Provided by University of Groningen

















