Which type of cell to become: Decision through indecision
From the moment of fertilization, building a human body involves a series of choices where cells generated by cell division must elect which of the myriad types of cell they will become. How does this decision occur? New research from Alice Moussy and colleagues of the Ecole Pratique des Hautes Etudes, Genethon and ENSLyon in France suggests that fate decision is not a unique programmed event, as was believed, but the outcome of a very dynamic process. The study, publishing July 27 in the open access journal PLOS Biology departs from the view that cell differentiation follows step-by-step instructions.
The majority of cells in the human body are blood cells, which comprise many different types that are continuously produced during the life of an organism. This variety is necessary to fulfil the diverse functions that blood performs—oxygen transport, immune defence or transport of nutrients, to name a few. All these very different cell types have the same ancestor, whose descendants must acquire different fates.
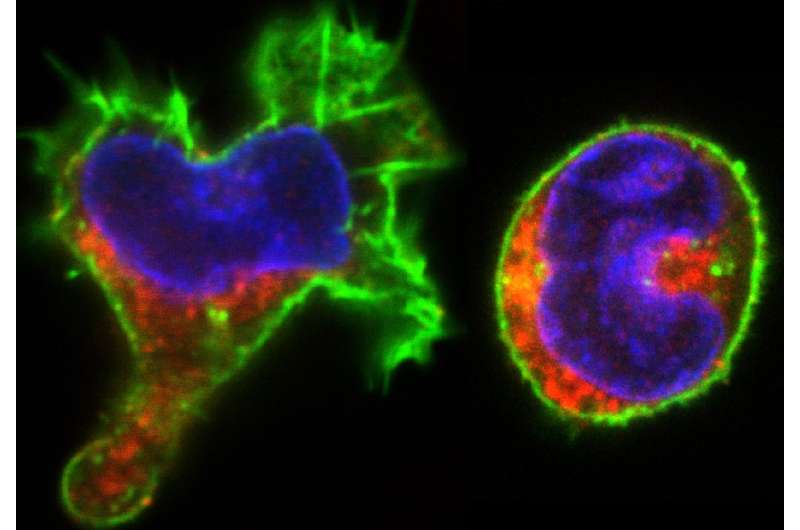
The blood ancestors are usually called hematopoietic stem cells; these cells were classically defined as a specific category of multipotent cells at the top of the hierarchy that can differentiate into any blood cell type by following a deterministic program. The authors used a time-lapse video recording coupled to single-cell molecular analyses to study the process step-by-step and found that fate decision is not a unique programmed event, but the result of a very dynamic process composed of spontaneous fluctuation and selective stabilisation of alternative cellular states.
The whole process is reminiscent of trial-and-error learning in which each cell explores—at its own rhythm and independently of cell division—different molecular possibilities (i.e. different genes turned on or off) before reaching a stable combination of active genes and the corresponding morphology. The authors observed that some cells seem to "hesitate" and change morphology many times before reaching a stable state.
Moussy and colleagues conclude that rather than being a feature of a specific cell category, multipotency appears to be a transitory state that can be reached back and forth by many different cells, driven by a complex network of dynamic molecular interactions.
More information: Moussy A, Cosette J, Parmentier R, da Silva C, Corre G, Richard A, et al. (2017) Integrated time-lapse and single-cell transcription studies highlight the variable and dynamic nature of human hematopoietic cell fate commitment. PLoS Biol 15(7): e2001867. doi.org/10.1371/journal.pbio.2001867
Journal information: PLoS Biology
Provided by Public Library of Science