May 4, 2017 report
Using a nickel catalyst with hydrocarbons to make fatty acids
(Phys.org)—A team of researchers with the Barcelona Institute of Science and Technology has developed a way to make fatty acids using a nickel catalyst along with hydrocarbons. In their paper published in the journal Nature, the group describes their technique and the uses to which it might be put. Matthew Gaunt and Patrick Williamson with the University of Cambridge offer a News & Views piece on the work done by the team in the same journal issue.
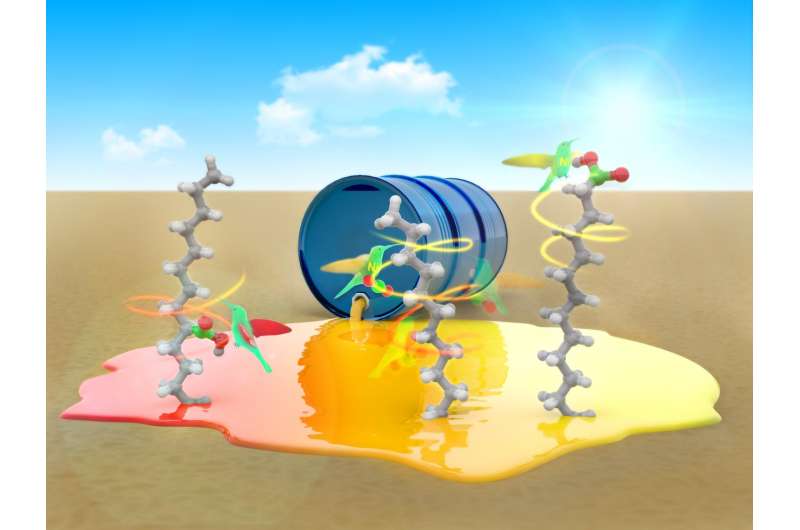
Fatty acids are used to make a wide variety of products, from plastics to soaps, rubber, drugs and food. They are long-chain compounds which are generally easy to make from animal lipids. But the end results typically have differing chain lengths, which requires purifying. Current processes used to make them generally suffer from several factors that can drive up costs, such as the creation of hazardous byproducts and the need for precious metals. In this new effort, the researchers have found a way to use carbon dioxide and hydrocarbons (isomeric alkenes) to produce single fatty acids in highly specific ways.
The new technique involves using a nickel catalyst that can be made to "walk" along a hydrocarbon chain, allowing for carbon dioxide to be incorporated at a desired reaction site. The two-step process begins with exposing the alkenes to hydrobromic acid, which adds bromine and hydrogen atoms across the carbon-to-carbon bond. The second part involves inserting a nickel atom into the carbon-bromine bond, which results in the formation of a carbon-nickel bond. The interactions that follow allow for the nickel to be walked along the chain until it reaches a spot where no interactions are occurring. The bond then reacts with carbon dioxide to form a single carboxylic acid product. The team notes that varying the temperature at which the reaction occurs allows for controlling how the reaction plays out including reversing the chain migration direction.

The advantage of the new technique is that it allows for using readily available materials and opens the door to the possibility of finding reagents other than carbon dioxide that could be used in similar reactions to create products made from alkenes.
More information: Francisco Juliá-Hernández et al. Remote carboxylation of halogenated aliphatic hydrocarbons with carbon dioxide, Nature (2017). DOI: 10.1038/nature22316
Abstract
Catalytic carbon–carbon bond formation has enabled the streamlining of synthetic routes when assembling complex molecules. It is particularly important when incorporating saturated hydrocarbons, which are common motifs in petrochemicals and biologically relevant molecules. However, cross-coupling methods that involve alkyl electrophiles result in catalytic bond formation only at specific and previously functionalized sites. Here we describe a catalytic method that is capable of promoting carboxylation reactions at remote and unfunctionalized aliphatic sites with carbon dioxide at atmospheric pressure. The reaction occurs via selective migration of the catalyst along the hydrocarbon side-chain with excellent regio- and chemoselectivity, representing a remarkable reactivity relay when compared with classical cross-coupling reactions. Our results demonstrate that site-selectivity can be switched and controlled, enabling the functionalization of less-reactive positions in the presence of a priori more reactive ones. Furthermore, we show that raw materials obtained in bulk from petroleum processing, such as alkanes and unrefined mixtures of olefins, can be used as substrates. This offers an opportunity to integrate a catalytic platform en route to valuable fatty acids by transforming petroleum-derived feedstocks directly.
Journal information: Nature
© 2017 Phys.org