March 31, 2017 report
Using platinum-molybdenum carbide to catalytically release hydrogen to power a fuel cell
(Phys.org)—A team of researchers from several institutions in China and the U.S. has developed a way to use platinum–molybdenum carbide to catalytically release hydrogen from methanol and water to power a hydrogen fuel cell. In their paper published in the journal Nature, the team describes the new method to produce hydrogen for possible use in a fuel cell.
As the planet continues to heat up, scientists around the world seek ways to power automobiles in a way that are as economical as gasoline. Such efforts have led to electric vehicles, hybrids, cars and trucks running on natural gas, ethanol and other fuels, and of course, the ever-elusive hydrogen fuel cell. In this new effort, the researchers suggest they may finally have found a way to make the last option viable.
Currently, there are a number of ways to obtain hydrogen for use in fuel cells, but thus far, none of them have proved economical enough to supplant the use of gasoline as the primary fuel for automobiles around the globe. In this new effort, the researchers suggest they may have come up with a process that could make hydrogen fuel cell vehicles more practical.
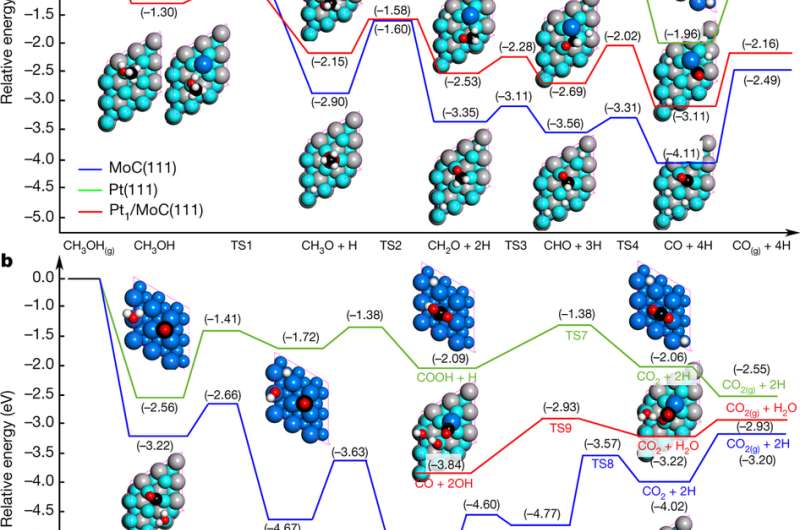
The process involves using a new catalyst, platinum–molybdenum carbide, to drive a reaction that results in the production of H2 and releases carbon dioxide. The team reports that the process can be done at temperatures from 150 to 190 C° and avoids the use of caustic materials. They claim it is five times as efficient as other techniques that use methanol. They also claim that a car with a 50 liter tank of methanol and just six to 10 grams of their catalyst could power a Toyota Mirai for approximately 690 km. Also, it would cost just $15 for the methanol and $320 for the platinum, which the team suggests, might be recyclable.
The group acknowledges that a process that releases carbon dioxide is not ideal, but note that many hydrogen-producing industrial processes do so, as well. They acknowledge that platinum is extremely expensive, but point out that current catalytic converters have approximately one to four grams of recyclable noble metals that could conceivably provide a source.
More information: Lili Lin et al. Low-temperature hydrogen production from water and methanol using Pt/α-MoC catalysts, Nature (2017). DOI: 10.1038/nature21672
Abstract
Polymer electrolyte membrane fuel cells (PEMFCs) running on hydrogen are attractive alternative power supplies for a range of applications1, 2, 3, with in situ release of the required hydrogen from a stable liquid offering one way of ensuring its safe storage and transportation4, 5 before use. The use of methanol is particularly interesting in this regard, because it is inexpensive and can reform itself with water to release hydrogen with a high gravimetric density of 18.8 per cent by weight. But traditional reforming of methanol steam operates at relatively high temperatures (200–350 degrees Celsius)6, 7, 8, so the focus for vehicle and portable PEMFC applications9 has been on aqueous-phase reforming of methanol (APRM). This method requires less energy, and the simpler and more compact device design allows direct integration into PEMFC stacks10, 11. There remains, however, the need for an efficient APRM catalyst. Here we report that platinum (Pt) atomically dispersed on α-molybdenum carbide (α-MoC) enables low-temperature (150–190 degrees Celsius), base-free hydrogen production through APRM, with an average turnover frequency reaching 18,046 moles of hydrogen per mole of platinum per hour. We attribute this exceptional hydrogen production—which far exceeds that of previously reported low-temperature APRM catalysts—to the outstanding ability of α-MoC to induce water dissociation, and to the fact that platinum and α-MoC act in synergy to activate methanol and then to reform it.
Journal information: Nature
© 2017 Phys.org