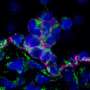
Monitoring cell fates
An international team of researchers led by ETH scientists has been studying the factors influencing the development of different blood cells. Their research shows that certain molecular mechanisms are not as relevant as previously assumed. This finding helps to improve our understanding of diseases such as leukemia and anemia.
In biological terms, a cell is the smallest functional entity of living organisms. The human body contains an enormous number of cells: somewhere in the region of 10 to 100 trillion, depending on a person's size and weight. Most of these cells perform specific functions in the body and are called differentiated cells. Stem cells, on the other hand, are able to continuously divide to produce more stem cells and differentiated cells, thereby providing an endless supply of differentiated cells. Certain cells in the body have a relatively short life span. For example, many white blood cells (leucocytes) and blood platelets (thrombocytes) die within a few hours to a couple of days, while red blood cells (erythrocytes) survive around four months.
Stem cell regeneration
Stem cells in the bone marrow thus produce millions of new blood cells every second. These stem cells are multipotent, which means they can generate all types of specialised blood cell with different functions: red blood cells, responsible for oxygen transport, white blood cells that are part of the body's immune defence system, and blood platelets which play a key role in blood-clotting. Exactly how stem cells develop into different cell types is still only partly understood. The process of differentiation - in other words, the decision as to which type of cell will be produced - depends on a number of different external and internal factors.
Timm Schroeder, Professor at the ETH Zurich Department of Biosystems Science and Engineering based in Basel, and his colleagues are studying the factors that play a role in the development of the individual blood cells. "The regulation of stem cell differentiation plays a vital role in maintining the normal process of blood formation", explains Professor Schroeder. "If this system starts to malfunction, it can lead to life-threatening diseases such as anaemia and leukaemia. We therefore need to have a better understanding of the molecular mechanism involved in this regulation."
Observation at the molecular level
The cell biologist and his team are analysing how stem cells differentiate into the different types of blood cell and how molecules in the cell nucleus (transcription factors) control this complex process. Working with the Helmholtz Zentrum Munich (German Research Centre for Environmental Health), they have developed an innovative microscopy technique for observing cells - cutting-edge equipment that is only found in very few of the world's stem cell research laboratories.
The two proteins GATA1 and PU.1 have been a particular focus of the researcher's attention. They play an important role in the differentiation of blood cells, explains Timm Schroeder. "They are transcription factors capable of activating or disabling comprehensive genetic programs with many target genes. This makes them powerful regulators of cell fates."
Promising potential
Using time-lapse microscopy, the researchers could observe living blood stem cells with unprecedented precision as they differentiated, while also quantifying the two proteins GATA1 and PU.1. "For decades it was thought that these two transcription factors were responsible for making the lineage decisions for stem cells. Now we are able to show that this is not the case, but that other mechanisms must be responsible for these decisions", explains Professor Schroeder. Research now needs to concentrate on other molecular mechanisms in order to understand the extremely complex process of blood stem cell differentiation.
Blood diseases such as leukaemia are severe disorders of the blood system. To improve our understanding of such diseases in future and to come up with effective treatments, we need to know exactly how the individual blood cells are created. A foundation stone for this research has now been laid at ETH Zurich.
More information: Philipp S. Hoppe et al, Early myeloid lineage choice is not initiated by random PU.1 to GATA1 protein ratios, Nature (2016). DOI: 10.1038/nature18320
Journal information: Nature
Provided by ETH Zurich