High-purity metals recovered from battery waste
Researchers at Lappeenranta University of Technology (LUT) have succeeded in recovering important metals – lithium, cobalt and nickel – from battery waste with nearly 100 percent purity. Recovered metals will be needed increasingly in the manufacture of batteries, and in the future especially in batteries of electric vehicles.
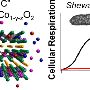
Researchers have been able to extract cobalt at 99.6 percent purity, nickel at 99.7 percent purity, and lithium at 99.9 percent purity from battery waste. According to Post-doctoral Researcher Sami Virolainen, who conducted the study, the purity of metals used as raw material in battery manufacturing is particularly important.
"The manufacturing of new batteries requires metals of particularly high purity. If the purity of lithium is below 99.5 percent, it is not suitable as raw material for batteries. In other words, the difference between 99.4 percent and 99.9 percent purity is very significant."
Lithium and cobalt are increasing in importance because they are needed in batteries of electronic devices and especially in electric cars. Forecasts indicate that the global need for lithium may quadruple between the years 2011 and 2025. Europe has few primary lithium resources, which means extraction and recovery from secondary raw materials are important ways to secure its availability.
"Lithium has not been labelled by the EU as a critical raw material, and it is not particularly expensive. However, changes in demand make it a raw material of considerable societal interest," reflects Virolainen.
Used batteries compose a potential raw material source for lithium recovery constantly increasing in quantity. Moreover, the operating life of batteries is only roughly a decade. In other words, batteries keep accumulating, but so does recyclable metal from battery waste.
The study separated metals through a liquid-liquid extraction process on a pilot scale. In the process, extraction takes place between two liquid phases which do not dissolve in each other. First, all other impurities are separated from the solution, leaving only lithium, cobalt and nickel. Previous studies have attempted to extract all three metals, but have only succeeded in extracting two at a relatively high purity and the third with low purity.
"As a separation process, liquid-liquid extraction is a viable option when nearly 100 percent purity and a high recovery rate are required."
Of the three metals, nickel is used in lower quantities for manufacturing batteries, and its availability is not at risk. However, in nature it is always associated with cobalt, and the two are difficult to separate. For use in batteries, cobalt must be separated from nickel. A high nickel recovery rate can be seen in the purity of the two other elements.
The research was conducted with a device with high flow rates simulating the industrial scale. The results of the research were presented in the Circular Materials conference organised by Chalmers University of Technology in early May.
Provided by Lappeenranta University of Technology