For 2-D boron, it's all about that base
Rice University scientists have theoretically determined that the properties of atom-thick sheets of boron depend on where those atoms land.
Calculation of the atom-by-atom energies involved in creating a sheet of boron revealed that the metal substrate - the surface upon which two-dimensional materials are grown in a chemical vapor deposition (CVD) furnace - would make all the difference.
Theoretical physicist Boris Yakobson and his Rice colleagues found in previous work that CVD is probably the best way to make highly conductive 2-D boron and that gold or silver might be the best substrates.
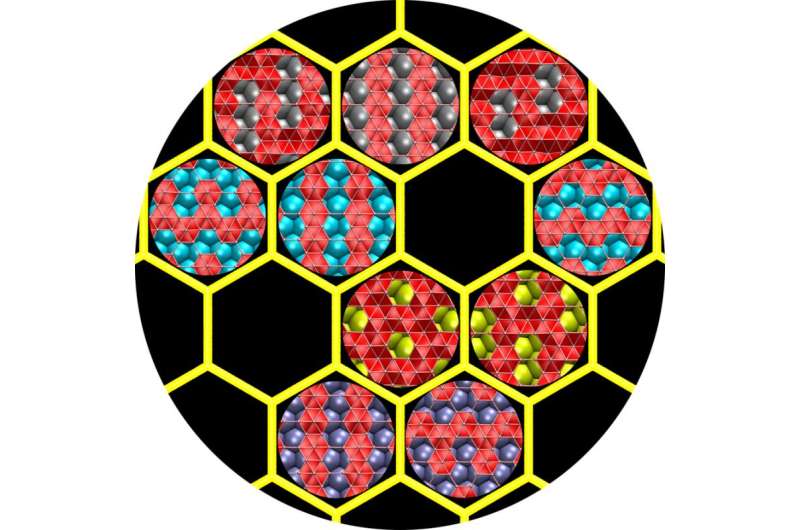
But their new calculations show it may be possible to guide the formation of 2-D boron by tailoring boron-metal interactions. They discovered that copper, a common substrate in graphene growth, might be best to obtain flat boron, while other metals would guide the resulting material in their unique ways.
The Rice team's results appear today in the journal Angewandte Chemie.
"If you make 2-D boron on copper, you get something different than if you made it on gold or silver or nickel," said Zhuhua Zhang, a Rice postdoctoral researcher and lead author of the paper. "In fact, you'd get a different material with each of those substrates."
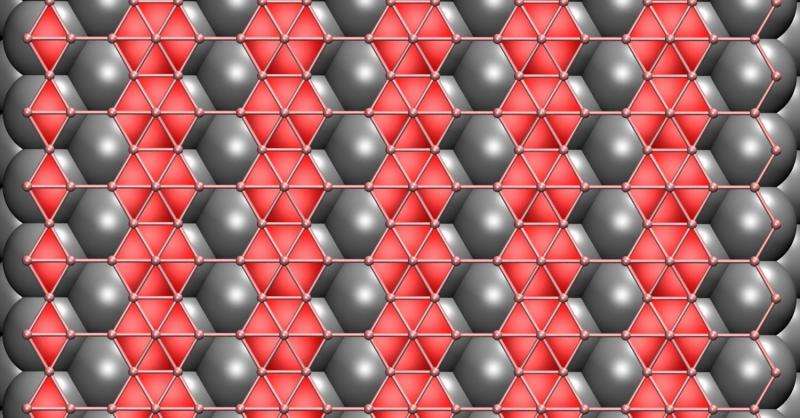
In chemical vapor deposition, heated gases deposit atoms on the substrate, where they ideally form a desired lattice. In graphene and boron nitride, atoms settle into flat hexagonal arrays regardless of the substrate. But boron, the researchers found, is the first known 2-D material that would vary its structure based on interactions with the substrate.
Perfectly flat boron would be a grid of triangles with occasional hexagons where atoms are missing. The researchers ran calculations on more than 300 boron-metal combinations. They found the pattern of atoms in a copper surface match up nicely with 2-D boron and the strength of their interactions would help keep the boron flat. A nickel substrate would work nearly as well, they found.
On gold and silver, they determined weak atomic interactions would allow the boron to buckle. In an extension, they theorized that naturally forming, 12-atom icosahedrons of boron would assemble into interconnected sheets on copper and nickel, if the boron supply were high enough.
One remaining downside to 2-D boron is that, unlike graphene, it will remain difficult to separate from its substrate, which is necessary for use in applications.
But that strong adhesion may have a side benefit. Further calculations suggested boron on gold or nickel may rival platinum as a catalyst for hydrogen evolution reactions in applications like fuel cells.
"In 2007 we predicted the possibility of pure boron fullerenes," Yakobson said. "Seven years later, the first one was observed in a laboratory. This time, with the enormous attention researchers are giving to 2-D materials, I'd hope some lab around the world will make 2-D boron much sooner."
More information: Angewandte Chemie, onlinelibrary.wiley.com/doi/10 … e.201505425/abstract
Journal information: Angewandte Chemie
Provided by Rice University