Self-healing material could plug life-threatening holes in spacecraft
For astronauts living in space with objects zooming around them at 22,000 miles per hour like rogue super-bullets, it's good to have a backup plan. Although shields and fancy maneuvers could help protect space structures, scientists have to prepare for the possibility that debris could pierce a vessel. In the journal ACS Macro Letters, one team reports on a new material that heals itself within seconds and could prevent structural penetration from being catastrophic.
It's hard to imagine a place more inhospitable to life than space. Yet humans have managed to travel and live there thanks to meticulous engineering. The International Space Station, equipped with "bumpers" that vaporize debris before it can hit the station walls, is the most heavily-shielded spacecraft ever flown, according to NASA. But should the bumpers fail, a wall breach would allow life-sustaining air to gush out of astronauts' living quarters. Timothy F. Scott and colleagues wanted to develop a backup defense.
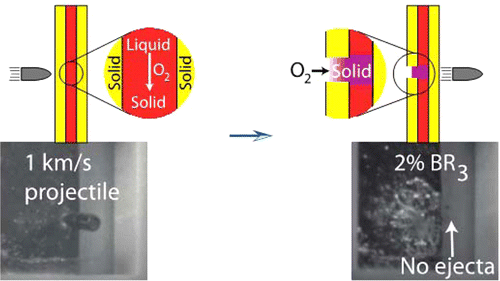
The researchers made a new kind of self-healing material by sandwiching a reactive liquid in between two layers of a solid polymer. When they shot a bullet through it, the liquid quickly reacted with oxygen from the air to form a solid plug in under a second. The researchers say the technology could also apply to other more earthly structures including automobiles.
More information: Rapid, Puncture-Initiated Healing via Oxygen-Mediated Polymerization, ACS Macro Lett., 2015, 4 (8), pp 819–824. DOI: 10.1021/acsmacrolett.5b00315
Abstract
Autonomously healing materials that utilize thiol–ene polymerization initiated by an environmentally borne reaction stimulus are demonstrated by puncturing trilayered panels, fabricated by sandwiching thiol–ene–trialkylborane resin formulations between solid polymer panels, with high velocity projectiles; as the reactive liquid layer flows into the entrance hole, contact with atmospheric oxygen initiates polymerization, converting the liquid into a solid plug. Using infrared spectroscopy, we find that formulated resins polymerize rapidly, forming a solid polymer within seconds of atmospheric contact. During high-velocity ballistics experiments, additional evidence for rapid polymerization is provided by high-speed video, demonstrating the immediate viscosity increase when the thiol–ene–trialkylborane resins contact atmospheric oxygen, and thermal imaging, where surface temperature measurements reveal the thiol–ene reaction exotherm, confirming polymerization begins immediately upon oxygen exposure. While other approaches for materials self-repair have utilized similar liquid-to-solid transitions, our approach permits the development of materials capable of sealing a breach within seconds, far faster than previously described methods.
Journal information: ACS Macro Letters
Provided by American Chemical Society


















