Long-distance solute transport in trees improved by intercellular pathways in living woody tissues
As large organisms, trees face some remarkable challenges, particularly regarding long-distance transport and communication. In addition to moving water and nutrients from their roots to their leaves, they must also integrate cell-to-cell communication over large areas. Furthermore, in order to function as a single, cohesive organism they must be able to effectively and efficiently send vital substances—such as DNA regulating signals—long distances along a network of cells, sieve-tubes, and vessels.
But how effective is this cell-to-cell communication and how far can such solutes travel?
Xylem and phloem serve as the two main arterial systems within a plant, and substances travel along two main pathways. Apoplasmic transport, in the system of cell walls, is important for movement of water and mineral solutes. Symplasmic transport, in which openings in the cell walls allow the cytoplasm of cells to be connected, facilitates the transport of nutrients, small ions, hormones, and transcription factors along a series of cells. Although this intercellular transport system is crucial for regulating cell differentiation and subsequent tissue maturation, it is not known how extensive these symplasmic pathways are and whether they can effectively transport solutes long distances.
The fact that woody tissues—specifically secondary xylem—contain long-living parenchyma cells, in addition to the dead cells primarily responsible for water transport, led Katarzyna Sokołowska and Beata Zagórska-Marek (University of Wroclaw, Poland) to investigate whether symplasmic pathways in these living cells might play an important role in transporting solutes from xylem to actively dividing tissues, such as the cambium. They've published their study in the most recent issue of the American Journal of Botany.
"Molecular transport of hormones and other signals in cell-to-cell communication is slow," notes Sokołowska. "Yet information is propagated quickly over large distances such as a tree height or width. Moreover, billions of cells building up a tree must quickly and efficiently communicate with one another. This is a necessity for maintaining this large organism's integrity throughout its development."
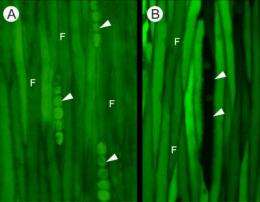
Sokołowska and Zagórska-Marek, in order to determine whether they could visually trace the pathways of solutes as those solutes travel up the living cells of the xylem network, cut 1- to 3-year-old branches from maple (Acer pseudoplatanus) and poplar (Populus tremula x P. tremuloides) trees and immersed the cut ends in a solution containing one of several fluorescent dyes.
After leaving the branches in the dye solutions for 3 to 7 days (adequate time for the dye to move a substantial distance), the authors cut cross- and tangential sections of the branches and found that dye was present in the living cells of both the radial and axial xylem parenchyma and ray and fusiform cambial cells—clear evidence that symplasmic transport occurred in both the secondary xylem and the cambial tissues. Moreover, dye was not only clearly seen in the cytoplasm of the cells, but also in the cytoplasmic bridges spanning walls between adjacent cells, confirming intercellular movement.
"Through the fluorescent marker application, we have successfully been able to visualize the transportation routes of nutrients or signaling molecules, which are regulated by changing states of cell-to-cell conductivity in time and in space," said Sokołowska. "They can be compared to traffic lights on crossroads, which control the road traffic and highway networks, ensuring fast and smooth transport between distant places."
Moreover, by using different dyes, the authors were able to see differences between the apoplasmic and symplasmic pathways. Although the apoplasmic dye was present in the cell walls of the secondary xylem, it was not visible in the cambial primary cell walls. This, together with the presence of the symplasmic dye in the cambial cells and in axial and radial xylem parenchyma, indicates the importance of the symplasmic pathway for transporting substances from the living xylem cells to the actively dividing cambial cells, which are crucial for developing new cells and tissues.
One of the most surprising and interesting finds was that the dye was not evenly dispersed throughout the cells despite the regular arrangement of rays in the cambium.
"We found that when fluorescent tracers are loaded to tree branches and migrate freely in the system of living cells, the tracer distribution becomes uneven," commented Sokołowska. "The regions appear with a high and low tracer content in the cells and they alternate along the branches. The pattern emerges in cambium, an embryonic tissue responsible for thickening of a tree through making its secondary conductive tissues, such as wood."
Father up from the source of the dye, the branch regions of rays filled with dye alternated with regions of rays that did not contain the dye—both dye and non-dye rays were composed of living cells with vigorous cytoplasmic streaming. The authors suggest that this pattern could be the result of differences in regulating the intensity of movement and unloading of dye from xylem to cambial cells in different areas.
"Most importantly, this regulation at the cellular level has a potential, through the spatial shift of the cellular states, of propagating important information in the whole population of interconnected plant cells over long-distances—similar to the green wave of streetlights for driving cars," Sokołowska stated.
In addition, this long-distance symplasmic movement from the xylem into the cambial rays is of utmost importance because the cambial cells are zones of active cell production and the intensively dividing cells in this region require many nutrients.
"Nowadays we are getting closer to understanding how cell-to cell communication can be translated into higher levels of plant body organization," Sokołowska concludes. "Cambium is a plant tissue composed of immortal, ever-dividing cells, equivalent to stem cells in animals. The more we learn about the mechanisms governing their functions, the better perspectives we get for directing morphogenetic processes in plants, animals and possibly humans."
"The wood produced by cambium is used commercially," she adds. "So, knowledge about cambial function and its regulation is also important for future modification of wood production quality."
More information: American Journal of Botany 99(12): 1745-1755. DOI: 10.3732/ajb.1200349
Journal information: American Journal of Botany
Provided by American Journal of Botany

















